Ethyl Acetate, also know as acetic ester, is an organic compound with the chemical formula C4H8O2. It is a kind of ester with functional group-Coor (double bond between carbon and oxygen). It can be used for common ester reactions such as alcoholysis, amolysis, transesterification, reduction, etc. It is mainly used as solvent, edible flavor, cleaning agent. HS CODE: 29153100
Ethyl acetate exhibits the following features:
Pleasant fruity odor: Ethyl acetate has a unique sweet fruity aroma, making it widely favored in various applications.
Volatility: Ethyl acetate is a volatile liquid that easily evaporates at room temperature, making it an ideal solvent in various industrial and laboratory applications.
Solvency: Ethyl acetate has excellent solvent properties and can dissolve various substances, including resins, oils, cellulose derivatives, and many synthetic polymers.
Low toxicity: Compared to many other organic solvents, ethyl acetate has lower toxicity, making it safer to handle and use.
Flammability: Ethyl acetate exhibits some flammability and should be handled with caution in the presence of ignition sources.
Biodegradability: Ethyl acetate can be biodegraded by microorganisms, reducing its environmental impact.
In summary, ethyl acetate possesses multiple advantageous properties and finds widespread applications in industries such as coatings, paints, adhesives, pharmaceuticals, and food processing.
| CAS # | EC Number | Hill Formula | Chemical Formula | Molar Mass |
| 141-78-6 | 205-500-4 | C4H8O2 | CH3COOC2H5 | 88.11 g/mol |
| Description | ||||
| Synonyms | Ethyl acetate, Ethyl ethanoate, Acetic ester, Ethyl ester | |||
| Ethyl Acetate Abbreviation | EAC | |||
| Product Information | ||||
| CAS number | 141-78-6 | |||
| EC index number | 607-022-00-5 | |||
| EC number | 205-500-4 | |||
| Hill Formula | C4H8O2 | |||
| Chemical formula | CH3COOC2H5 | |||
| Molar Mass | 88.11 g/mol | |||
| HS Code | 29153100 | |||
| Molecular Structure | 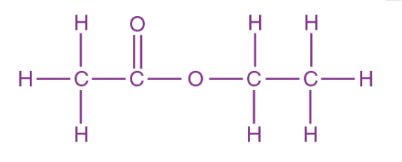 | |||
| Physicochemical Information | ||||
| Boiling point | 77 °C (1013 hPa) | |||
| Density (d 20 °C/ 4 °C) | 0.90 g/cm3 (20 °C) | |||
| Explosion limit | 2.1 - 11.5 %(V) | |||
| Flash point | -4 °C | |||
| Ignition temperature | 460 °C | |||
| Melting Point | -83 °C | |||
| Vapor pressure | 97 hPa (20 °C) | |||
| Solubility | 85.3 g/l | |||
For the package, Ethyl Acetate (EAC) is available in drum and isotank. The drum contains 180kg, and the container load for 20FCL is 14.4tons, 40FCL is 24.48tons with pallet and 25.2tons without pallet. The isotank contains 20-21MT.
Due to its flammability and volatility, Ethyl Acetate (EAC) may ignite and cause fires or explosions when exposed to high temperatures or ignition sources. Therefore, protective measures should be taken when storing and delivering ethyl acetate:
Ethyl Acetate (EAC) is Class 1 flammable substances and should be stored in a cool, ventilated place, away from sources of ignition.
Measures should be taken to prevent static electricity. During loading and unloading, handle with care to prevent damage to packaging and containers and to prevent the accumulation of static electricity.
The product should be stored in a cool, ventilated warehouse with a temperature not exceeding 30°C, and should be protected from direct sunlight. Containers should be kept tightly closed. Store separately from oxidants, acids, and alkalis.
The workplace should be well ventilated, and personnel should wear protective equipment.
Ethyl Acetate (EAC) has a wide range of applications across various industries due to its versatile properties. Some common applications of ethyl acetate include:
Solvent: Ethyl acetate is primarily used as Ethyl Acetate Solvent in the production of paints, coatings, adhesives, varnishes, and inks. Its strong solvency power makes it effective in dissolving various resins, polymers, and organic compounds.
Extraction: Ethyl acetate is utilized in the extraction of natural products, such as essential oils, flavors, and fragrances, from plant materials due to its ability to efficiently extract organic compounds.
Pharmaceuticals: Ethyl acetate is used in the pharmaceutical industry as a solvent in the manufacturing of drugs, pharmaceutical formulations, and as a carrier solvent in drug delivery systems.
Perfumes and Cosmetics: Ethyl acetate is commonly used in the formulation of perfumes, fragrances, and personal care products due to its pleasant fruity odor and low toxicity.
Nail Polish Remover: Ethyl acetate is a key ingredient in nail polish removers due to its ability to effectively dissolve nail polish without damaging the nails.
Laboratory Reagent: Ethyl acetate is frequently used as a solvent in chemical laboratories for various analytical and preparative procedures.
No, ethyl acetate and acetone are not the same. They are two different chemical compounds with distinct structures and properties. Ethyl acetate (CH3COOCH2CH3) is an ester compound commonly used as a solvent in various industrial applications such as paints, coatings, adhesives, and pharmaceuticals. It has a sweet, fruity odor and is flammable. Acetone (CH3COCH3) is a ketone compound often used as a solvent, particularly in nail polish removers, paint thinners, and industrial cleaning agents. It has a strong odor and is also flammable.
Ethyl acetate is generally considered a safe food additive and is confirmed as suitable for food by regulatory bodies such as the US Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA). It is often used as a spice in foods and is used in products such as sweets, pastries and drinks. However, like any other food additive, ethyl acetate should be used in accordance with the relevant regulations and within the prescribed limits to ensure its safety.
According to current scientific research, ethyl acetate is not considered a carcinogen. However, high concentrations of ethyl acetate may cause certain health effects, especially in the case of long-term or high exposure. Therefore, when using ethyl acetate, it is still necessary to follow the relevant safe operating procedures and ensure proper ventilation and personal protection measures.
Contact Us for More Information about Douwin Chemicals
Get in Touch
Email us with any questions or inquiries or use our contact data. We would be happy to answer your questions.